Continuous manufacturing mode for large-scale GMP production.

NANOus® represents the future of commercial-scale GMP manufacturing. With continuous operation and a flow rate up to 1200 mL/min, NANOus® removes the need for parallelization of production lines, significantly lowering operating costs for CDMOs and large pharmaceutical companies. NANOus® features a highly configurable closed system with features like process analytical technology (PAT) and cleaning and sterilization in place (CIP/SIP) to ensure every batch delivers the highest quality.

Key Benefits
→Continuous manufacturing customized to your process for reliable commercial production
One step scale-up from NANOlab®
Designed for Grade C aseptic manufacturing
Removes the need for parallelization on manufacturing lines
Continuous operation mode
Flow rates up to 1200 mL/min
Technical Specifications

| Application | Commercial-scale GMP production |
| Dimensions* | approx. 2.4 m x 1.0 m x 2.2 m |
| Total flow rate | <1200 mL/min |
| Batch size | No upper limit (continuous) |
| No. of inlets | 2 (3rd channel for ILD available) |
| Mixing technology | FR-JET® modular mixer |
| Process mode | Continuous |
| GMP capability | Full GMP, cleanroom-compatible |
| Single-use | All product-contacting parts |
| Integration | In-line dilution, TFF, sterile filtration |
*All dimensions are width x depth x height
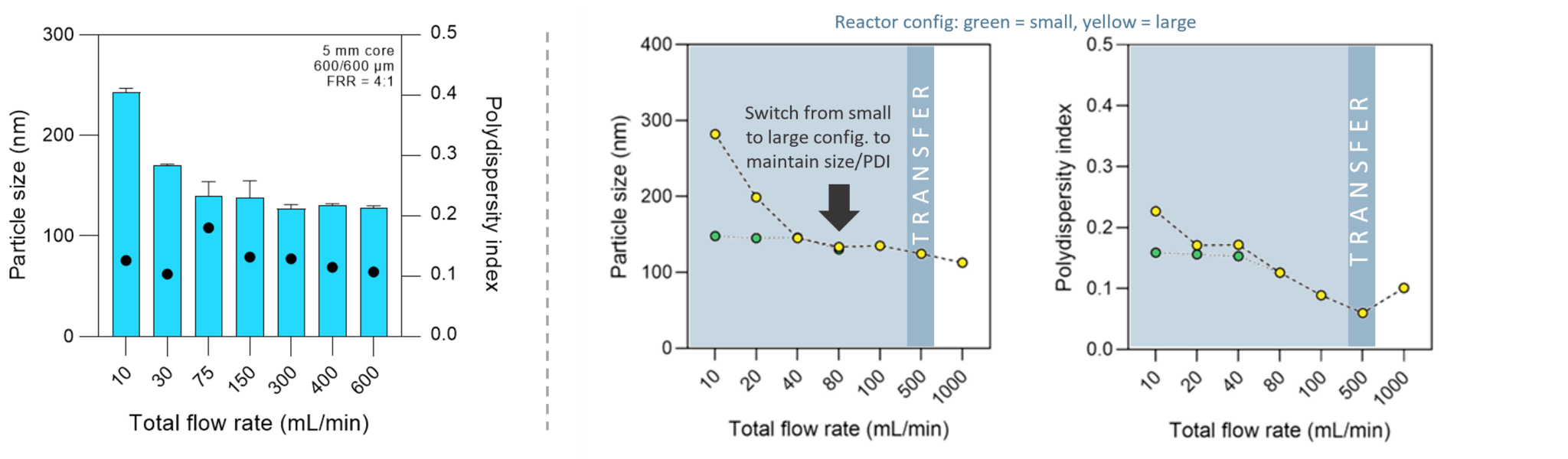
Request a QuoteExample data showing particle property control across scales
Nanoparticle size and polydispersity remain consistent as you scale from NANOlab® through to NANOus®, demonstrating true seamless scale-up with the FR-JET® modular mixing technology.
The NANOus® was developed in partnership with Harro Höfliger, a leading provider of technology solutions for pharmaceutical manufacturing, including aseptic processes.
Scale Without Limits
The NANOus® system is built for commercial-scale production, talk to us about your manufacturing requirements.
Request a Quote