Industry's first pumpless, fully disposable cassette-based equipment
for nanoparticle manufacturing.

The NANOme® GMP system reimagines your aseptic manufacturing experience. Pump-free, fully disposable cassette-based equipment enables Grade C aseptic manufacturing with a turnaround time of just 20 minutes between batches. With direct method transfer from our NANOlab® system, and no process re-development, you can de-risk your CMC program and eliminate the question "Can we scale to GMP?"

Key Benefits
→Seamless transition from non-clinical development to clinical grade material
Designed for small scale GMP batches, up to 1.2 L
Deliver 100+ batches per day
Pump-free, fully disposable cassette-based manufacturing
No cleaning required, batch-to-batch turnaround in 20 min
GAMP 5 & CFR 21 part 11 compliant software
The NANOme® System

Front view, fully enclosed system with integrated touchscreen

Doors open, disposable cassette and fluid path visible

Quick-swap cassette, 20-minute batch-to-batch turnaround
Technical Specifications

| Application | GMP clinical trial manufacturing |
| Dimensions* | approx. 70 cm x 45 cm x 90 cm |
| Total flow rate | 20 – 240 mL/min |
| Batch size | 50 – 1,200 mL |
| No. of inlets | 2 (3rd channel for ILD available) |
| Mixing technology | FR-JET® modular mixer |
| Process mode | Batch |
| GMP capability | GMP-ready, cleanroom-compatible |
| Single-use | All product-contacting parts |
*All dimensions are width x depth x height
Request a QuoteUnparalleled performance proven by data
The NANOme® delivers consistent process performance, high batch reproducibility, and extraordinary process stability, validated through rigorous testing and real-world manufacturing data.
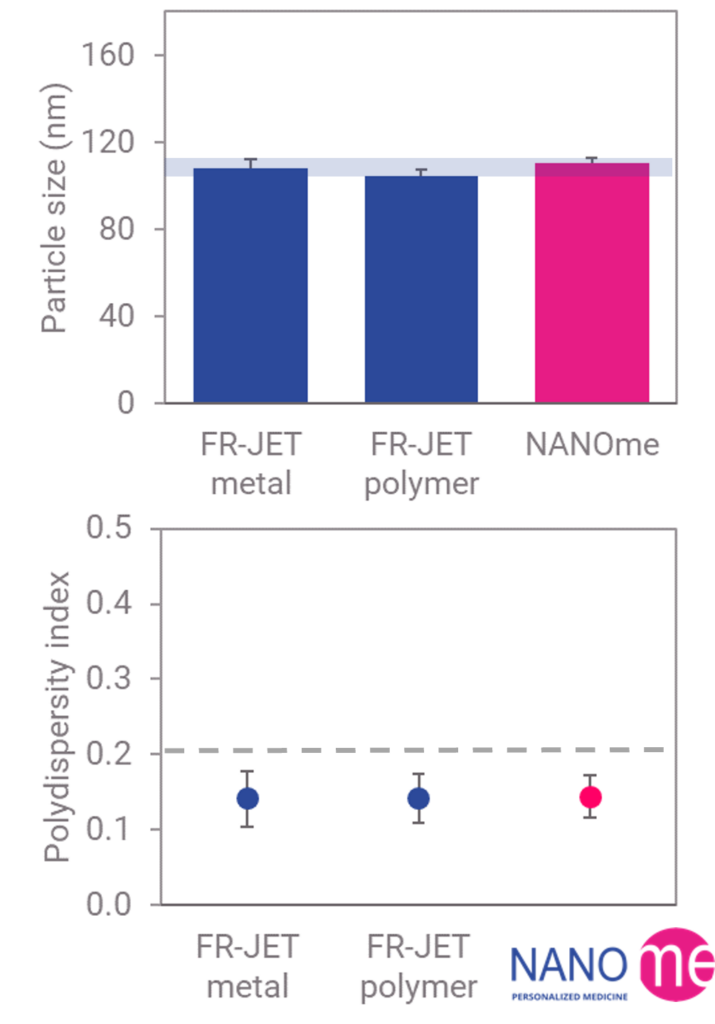
PLGA Nanoparticles
Consistent particle size (~110 nm) and low polydispersity index across FR-JET® metal, FR-JET® polymer, and NANOme® cassette systems.
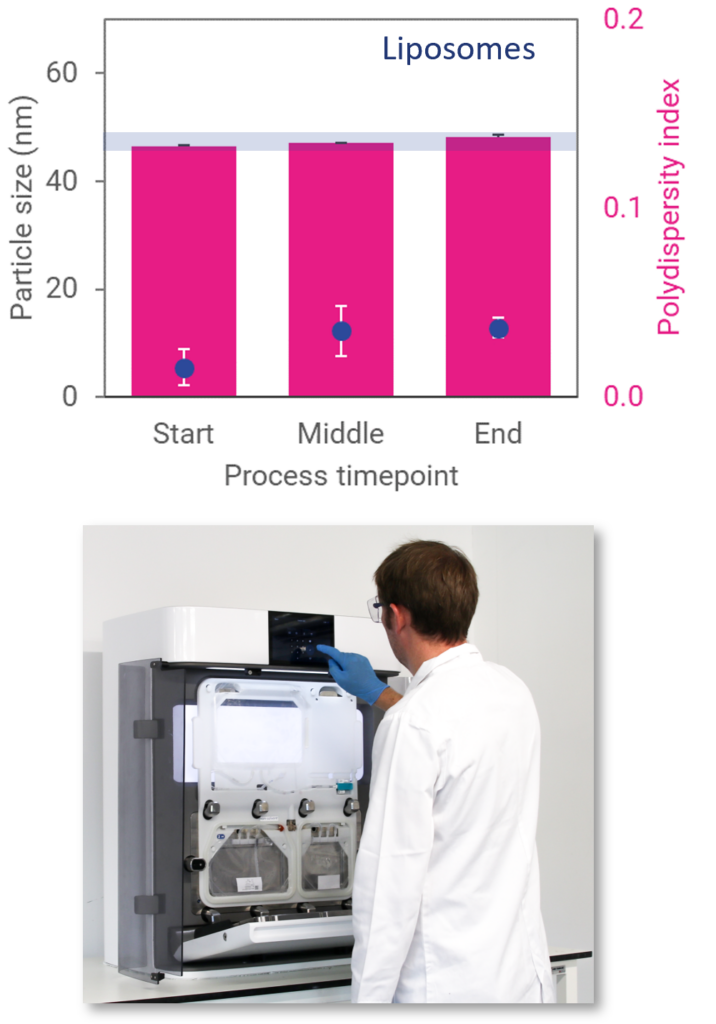
Process Stability
Liposome particle size remains stable throughout the entire batch, from start to middle to end, demonstrating exceptional process consistency.
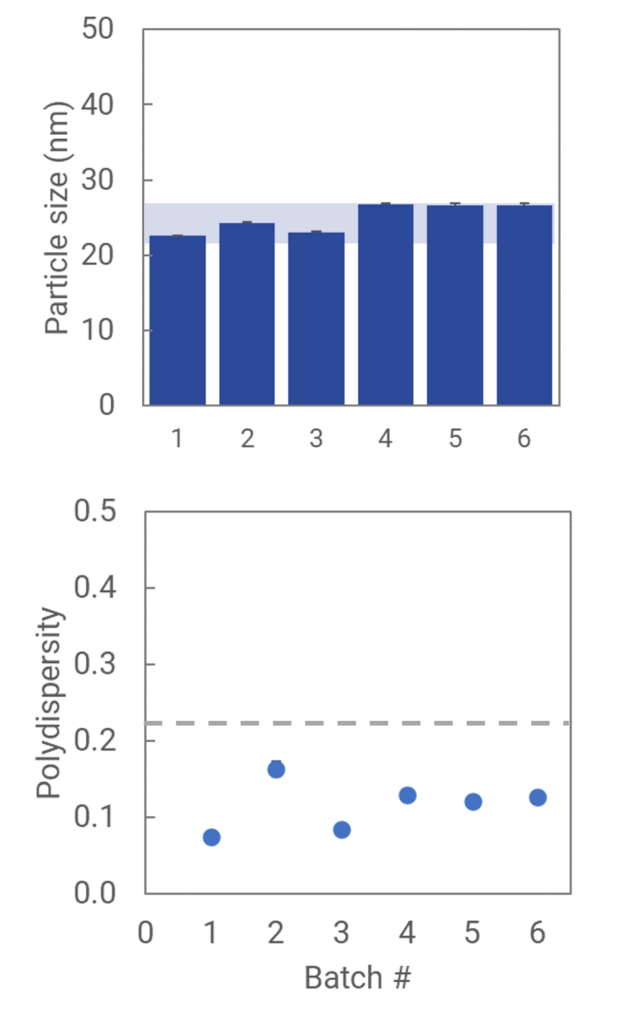
Batch Reproducibility
Six consecutive batches demonstrate tight particle size control (~25 nm) and consistently low polydispersity, proving batch-to-batch reliability.
Ready for GMP Manufacturing?
Our team can guide you through the transition from development to GMP-grade nanoparticle production.
Schedule a Consultation